Investigation of mass transport processes in a microstructured membrane reactor for the direct synthesis of hydrogen peroxide
Abstract
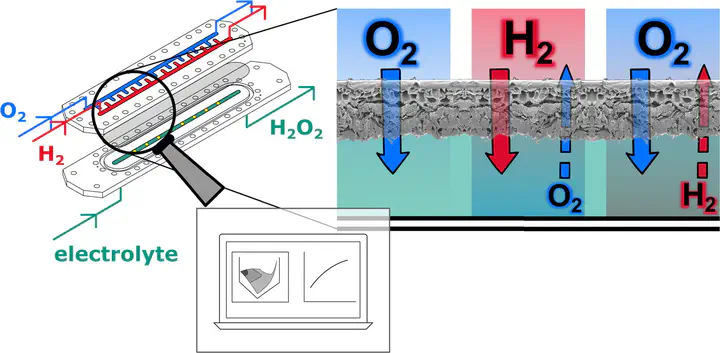
Microstructured membrane reactors present a promising approach to master the productivity and safety challenges during the direct synthesis of hydrogen peroxide. However, various mass transport processes occur in this complex system. In order to gain a deeper understanding of these processes, the saturation and desaturation behaviour of the liquid reaction medium with the gaseous reactants is investigated experimentally to examine possible cross-contamination. Moreover, the employed PDMS membrane’s permeances to hydrogen and oxygen are researched at different pressures, by using a variable-pressure/constant-volume setup for the behaviour at ambient pressure and a constant-pressure/variable-volume setup for the behaviour at elevated pressures. A mathematical model in MATLAB is applied to simulate the results. It is shown that a certain desaturation of the gasses through the membrane occurs, and the results are underlined by the modelled ones using a solution-diffusion model in MATLAB. Thus a constant flushing of the gas channels of the reactor is required for safety reasons. Moreover, the measured permeance values indicate that the species transport is mainly limited by the diffusion in the liquid phase and not the membrane resistance.